IPROVE Scope & Ambition
1. Why is a Vaccine R&D roadmap essential for Europe today?
There is no doubt that vaccination is one of public health’s most valuable pillars. Immunisation through vaccines has prevented more premature deaths, permanent disability, and suffering in all regions in the world, than any other medical intervention1. Without vaccination many of the health, and therefore economic and social gains of the past 200 years would simply not have been possible.
Vaccination has eradicated smallpox in man and rinderpest in cattle and it has dramatically reduced the burden of numerous infectious diseases, especially in infants and children2. It has freed societies and economies from a huge burden of childhood morbidity and mortality and underpinned individual and social growth, prosperity and wellbeing. In the coming decade, vaccines are projected to save 25 million more people3, and there is no doubt that both prophylactic and therapeutic vaccines with the potential to prevent, or even cure, communicable and non-communicable diseases will continue to be fundamental to public health in the future4.
Nevertheless, the demand for new and improved vaccines to address unmet and emerging medical needs requires the right technical and scientific skill set, sustained and sufficient investment, as well as, a structural framework that incentivises and rewards research and innovation. Strengthening partnership and coordination across relevant actors and sectors is as essential to vaccine innovation and supply as the underlying scientific and technical capabilities and capacity. A strategic and forward-looking approach is key. All of the challenges affecting the current vaccine and vaccination landscape in Europe require joint stakeholder reflection to ensure a coherent EU strategy for vaccination. The IPRove (Innovation Partnership for a Roadmap on vaccines in Europe) FP7-funded Coordination and Support Action5 was conceived to propose a roadmap for how Europe can best invest in the science and technology essential for vaccines innovation. The task was to cover all areas from discovery and development to production and uptake, as well as to initiate reflection on the political, legal, economic and structural measures that will best incentivise, reward and accelerate the development of vaccines. This roadmap is expected to guide and inform the future european research commitments and investment priorities in order to create an appropriate enabling environment to spur vaccine research, know-how, and innovation. The ultimate goal is to safeguard and advance public health in Europe and the world and ensure the competiveness of Europe in the area of vaccines, where the region has traditionally held a leadership position. Since vaccines are widely recognised as essential tools in maintaining public health, an adequately supportive and innovation-friendly R&D environment is absolutely critical to drive the development of novel vaccine technologies. This requires putting in place the appropriate “push” (e.g. capacity, capability, funding, tax incentives and infrastructure) and “pull” (such as priorities, attractive markets, stable demand, and favourable procurement policies) mechanisms. These need to be shepherded by the right processes, political will, and social and economic environment, allowing europe to stay at the leading edge of competitiveness for this strategic and vital health sector.
MEETING TODAY’S AND TOMORROW’S PUBLIC HEALTH CHALLENGES
Vaccination also has an important role to play in the global fight against the rising threat of antibiotic resistance. While the effective use of existing vaccines can help reduce the need for antibiotics or promote a more rational use, renewed research efforts targeting new generations of vaccines aimed to tackle antibiotic resistant bacteria and healthcare associated infections should be of primary importance6. With changes to the social and demographic structure of the population in the EU, there is a need to re-think the way we deliver healthcare. Increasingly we must focus on preventative approaches that will help individuals continue leading healthy lives. Currently, spending on health prevention represents less than 3% of the overall healthcare spending in the EU (OECD, 2013), and there is room for identifying more efficiencies to optimise health promotion and disease prevention programmes.In order to deliver such innovation, there is a need for tools that enable us to prioritise targets for vaccine innovation. Once this prioritisation is clear, the process of developing innovative vaccines requires considerable investments, and efforts to adapt existing or establish new breakthrough technologies. The process can take up to 20 years from R&D to availability on the market. Access to essential vaccinations must go beyond childhood. Indeed adults are no less valuable than children and a life- course approach to immunisation is required if we are to ensure equal access across all ages, geographies and societies. This calls for better implementation of currently available vaccines, but also for the innovative R&D of ‘personalised’ vaccines designed to meet specific target group needs in specific healthcare settings. Such an agenda requires putting in place the appropriate infrastructure that can help identifying population needs, but also understand the performance of current vaccination schemes, thus appropriately monitoring epidemiology, coverage, and effectiveness of the programmes in place.
EUROPE’S TRADITIONAL LEAD NEEDS TO BE SUSTAINED
Europe is a long-standing leader in both vaccines and public health. The first golden age of vaccination was led by the European pioneers of germ theory, pathogen culture and vaccines such as Pasteur, Koch, Ramon, Mérieux, Sclavo, Von Behring and others and the development of vaccines, which protected against rabies, diphtheria, tetanus, pertussis, and TB. This was accompanied by the establishment of national vaccine institutes in Europe and around the world, e.g. the Pasteur Institute, Wellcome, the Robert Koch Institute, and the Sclavo Institute.
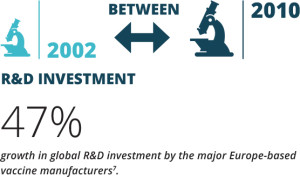
Expertise exists in Europe in most of the critical disciplines required for vaccines innovation. Europe is prominent in the field of adjuvants, and has also an excellent track record of inventing and exploiting novel routes of delivery. Beyond knowledge, cutting-edge critical mass exists in key areas such as infrastructures (e.g. animal facilities, BSL-3/4 containment facilities), and supporting technologies (bio- imaging, histo-pathology, tracing, immuno-monitoring). The know-how, regulatory framework and capacity to conduct clinical trials also constitute strong assets for Europe. Furthermore, most of the technology suppliers for vaccines manufacturing are based in Europe, ensuring access to state-of-the-art innovation. Even though existing figures demonstrate the capacity of European R&D to support vaccine innovation, the number of R&D projects in Europe has plateaued in the period 2002 to 2010 in favour of increased investments in emerging economies. Increasingly me-too and state-supported innovative vaccine development and production are taking place elsewhere in the world.

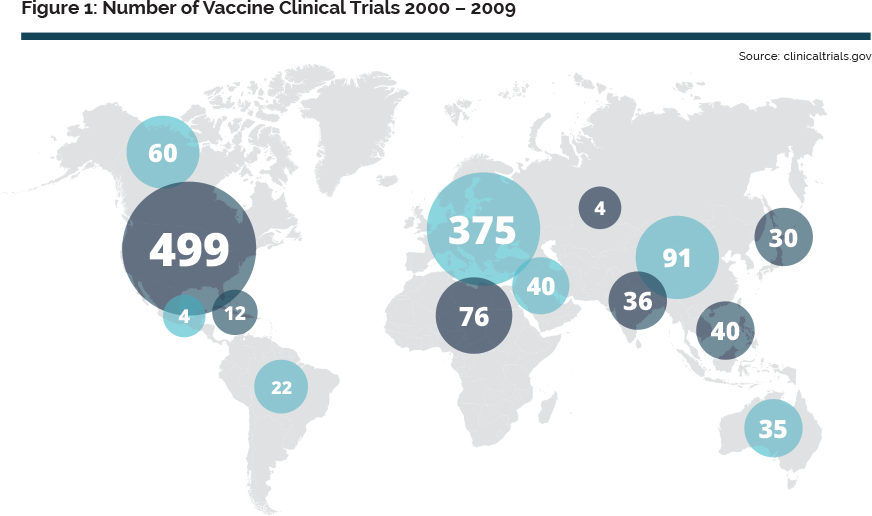
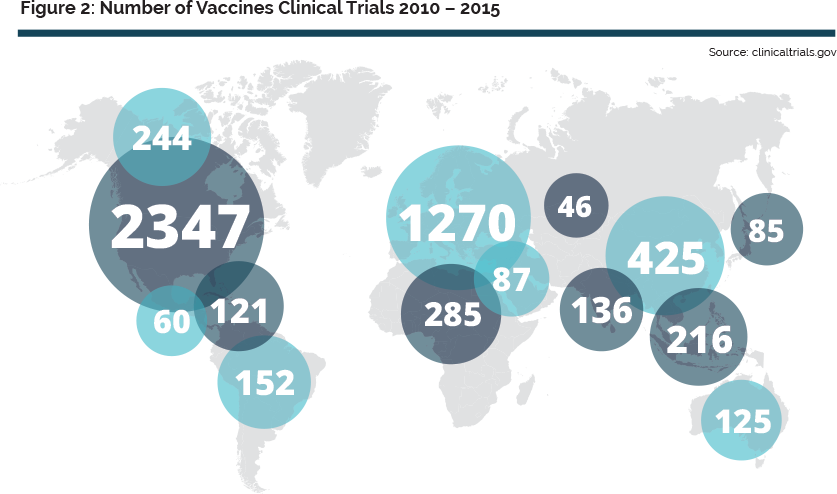
This shifting balance is a consequence of the broader recognition of the strategic social as well as economic value of vaccine production. Consequently many governments in emerging countries are investing heavily in developing domestic vaccine research, development and production in order to achieve the necessary critical mass and, more importantly, to secure access to vaccines. We currently witness the establishment or re-establishment of vaccine capability and capacity in countries such as China, India, Vietnam, Indonesia, Thailand, South Africa, Brazil, Mexico, Argentina, and elsewhere8. Keeping Europe’s lead in such a key sector requires a concerted and coordinated effort to better pool and leverage the capacity and capability distributed across europe in its centres of excellence. Vaccinology is intrinsically multi-disciplinary, and therefore functions most effectively if the necessary critical mass of capacity, capability, financing, translation and cooperation is reached and combined, particularly through co-localisation. This explains the concentration of vaccine-related critical mass in geographical clustering of vaccine research, development, venture capital, SMEs and large vaccine producers in the last decades. This has been most notable on the East and West coasts of the USA and is starting to develop in India (e.g. Hyderabad) and China (e.g. Shenzhen). By bringing together a broad range of critical stakeholders, IPROVE aims to build a comprehensive and clear overview of the gaps in investment, science and structure that, if filled, would reinvigorate eu vaccination leadership.



THE VIABILITY OF THE CURRENT VACCINE R&D MODEL IS AT RISK
Whilst recent innovation in vaccine research and development is widely acknowledged, the unique characteristics and challenges of vaccine R&D are less well recognised. Vaccines are distinctive from other medicinal products for at least two reasons; first, they are preventative in nature and, as such, intended for a larger number of healthy subjects and so must have an appropriate risk-benefit profile. Secondly, they are highly technical process-dependent biological products that are hard to characterise as finished products, so require strict process control to minimise their innate variability and unpredictability.These special features make the development and production of vaccines particularly time-consuming, demanding, complex and costly and expose it to numerous risks. Even the development in very early stages, such as exploratory and pre-clinical research and development, to simply identify a candidate vaccine can cost up to us $20 million.
In addition, increasing hurdles on the demand side are undermining the competitiveness of the vaccine sector as a whole and its ability to continue investing in innovative R&D. Growing trends of low price driven procurement policies fail to recognise the cost of quality, supply reliability and innovation of vaccines. Health Economic Analyses (HEA) fail to factor in broader and indirect economic and societal benefits of vaccines. There is certainly a need to foster the development of appropriate and comprehensive evaluation of frameworks that are adapted to the specificities of vaccines and immunisation in general. In particular, it is thought that the EU could benefit from further developing the network of existing National Immunisation Technical Advisory Groups (NITAGs), and improving coordination and expertise with Health Technology Assessment (HTA) bodies. This is in the interest of citizens’ public health. It is also in their interest to incentivise R&D efforts in vaccines, fully capitalising on the complexity of their research and manufacturing.
Unlike other pharmaceuticals, vaccine development can take up to 15 – 20 years without counting time to effective population access (on average a median time-lag of an extra 6.4 years9 after marketing authorisation). A vaccine may require clinical testing in 15-20 times as many subjects as for pharmaceutical drugs10 and costing up to US $ 900 million per vaccine production unit11.
The manufacturing itself is a complex and lengthy process. 6 to 24 months may elapse between the vaccine being available in bulk form and it being distributed, with 70% of the production times consumed by quality control. Opening and qualifying a new production facility may take more than 5 years and represents colossal investments. While the average cost of a single biological manufacturing site depends on its location and product, the cost can range from US$ 100 million to 600 million dollars12 or more.
VACCINE RESEARCH IN EUROPE NEEDS CONNECTING
The panel participants in the IPROVE consultation voiced the opinion that the European vaccine field could be better interconnected. They felt that the sub-optimal fragmentation may be driven by the fact that European countries have their own national vaccination decision-making processes. As a result, whilst Europe is a single economic community it is not a single or even interconnected public health or vaccination community. Indeed in some EU countries there are as many vaccination programmes as there are autonomous states/regions (e.g. Spain). This separation of economic and public health unity is driven by the European principle of subsidiarity in healthcare in Europe. The result is an inevitable heterogeneity of vaccination programmes and associated diversity of vaccine priorities and vaccine R&D focus and funding. This means that while there are many centres of vaccine excellence around Europe, their sharing of agendas, expertise, experience and personnel is limited.
Sub optimally connected European vaccine R&D programmes reduce the capacity to compete or collaborate with the huge critical mass of vaccines and vaccination R&D found in clusters such as Cambridge MA, USA. The panel therefore proposed that this apparent fragmentation should be addressed through an appropriate mix of push and pull mechanisms as well as being shepherded by the right processes, political will, and social and economic environment. Some reviewers have nevertheless observed that the postulated sub-optimal integration of Europe’s vaccines R&D capability and capacity is not evidence- based. It may, therefore, be appropriate for the EU to conduct a baseline study to check this assumption and if confirmed to track the progress of future initiatives to improve it.
2. IPROVE approach
IPROVE is a first attempt to develop a holistic view on this important sector as no similar project with a European scope has ever been funded by the EU’s programme. Three main features contribute to the uniqueness of the IPROVE initiative.
TAKING A CROSS-CUTTING CAPABILITIES, CAPACITIES RATHER THAN A DISEASE-BASED APPROACH
IPROVE focused on building critical stakeholder consensus on the priority gaps and challenges as well as recommendations for eu-level action on common topics of interest to bolster vaccine innovation across the entire innovation chain. The consortium considered that certain disease-based approaches have already been funded and explored to some extents with the support of existing and previously funded initiatives at national, EU, and international levels, including based on the publication of the recently updated WHO Report on Priority Medicines for Europe13. Therefore, the project concentrated efforts on technologies and cross cutting horizontal bottlenecks that must be overcome to allow the delivery of new generation vaccines in each of the key areas of unmet medical needs.
It is thought that this could help to support a more targeted approach in the allocation of funding, by strategically investing in the most promising and forward- looking partnership models. Efforts should be directed towards projects and players with higher potential for delivering true innovation in order to clearly meet the health, demographic change, well-being and security challenges of today, in line with the goals of the eu multi-annual financial framework.
ALIGNING AND PRIORITISING ON FUTURE EU EFFORTS
Much of what emerged from the IPROVE consultation process confirms and builds on research initiatives already happening across different countries in Europe. These however lack the intent for pan-European coordination and alignment across the different sets of stakeholders at EU level that should help their implementation.
Therefore, the added value of IPROVE is that it allowed different sets of stakeholders to share their ideas on what should be on top of the vaccine R&D agenda, align their thoughts, and finally prioritise on the main focus areas for EU intervention, with potential for cross-fertilisation. Through this prioritisation, the aim is to help policy-makers and funders identify the most relevant technologies where investment is key in the short and medium term.
FOSTERING BETTER INTERACTION BETWEEN EU STAKEHOLDERS
Europe currently benefits from the presence of a core set of more or less formalised national and regional vaccine ‘clusters’ in some EU Members States, such as France (Lyon Biopôle), UK (UK Vaccines R&D Network), Belgium, Italy, and the Netherlands. Building on this environment, IPROVE created the momentum and gave impetus to encourage increased interaction not only within but also across such clusters. This should help laying the foundation for favouring the establishment of an actual European working network across disciplines and experts between public and private, but also public-to-public and private-to-private players. IPROVE should be regarded as a first milestone in the above directions, and the success of its ambition will depend on stakeholder ownership and responsibility to implement its recommendations, particularly at the EU political level. Furthermore, though agreement on the content of such recommendations exists, more coordination and investment will be needed as to better understand what the best models of collaboration and financing mechanisms are.
3. IPROVE methodology
The IPROVE methodology allowed a broad and comprehensive stakeholder consultation, with a view to increasing chances that the resulting roadmap is holistic, and realistic, and able to meaningfully inform structures, processes, projects and funding that truly address the shared needs across public and private vaccine research at both national and EU level. This bottom-up stakeholder consultation process was structured around six pillars/thematic areas. Each pillar was the subject of a specific stakeholder consultation workshop. The pillars or thematic areas identified were: 1) Vaccine R&D, 2) manufacturing and quality control, 3) infrastructure, 4) therapeutic vaccines, 5) SMEs, 6) vaccines acceptance and training needs.
Each workshop aimed to engage a range of high-level experts whose roles and expertise matched the scope and breadth of the topic and roadmap. Participants represented stakeholders from across the entire vaccine and vaccination innovation spectrum, from industry, academia, and public health to national vaccine research institutes, international organisations, and funding agencies. Furthermore, experts of the European Commission and regulatory agencies were invited and participated in the workshops to complement the external stakeholder engagement.
All of the stakeholder consultation workshops were aimed to achieve the following objectives:
- Identify common critical gaps, fragmentation and silo challenges affecting each of the specific areas tackled during the consultation
- Provide specific theme-based recommendations where consensus could be found on priority areas for investment through EU and national funding programmes
- Describe changes in framework conditions that would help drive, incentivise and facilitate prioritised vaccines R&D in Europe in the defined topic areas dealt with.
A first draft roadmap resulting from this broad consultation was submitted to three main groups of stakeholders for iterative consultation, in order to refine the priorities and recommendations:
- The IPROVE consortium partners, inclusive of their respective networks of experts
- The IPROVE Advisory Board of four independent scientific experts14 and
- The IPROVE Affiliate Members Group, comprising of leading vaccine experts and representatives of public authorities in charge of vaccine programmes and funding at national level15.
The draft roadmap was finally put to further stakeholder consultation through a publicly accessible web-based platform targeting the leading exponents of the vaccine community in Europe that participated in the workshops16. This roadmap is intended and expected to be of use to the European Commission and relevant national and regional institutes and institutions operating at the Member State level, in view of informing their funding programmes.
4. Limitations
The IPROVE Consortium believes that the following main limitations should be duly considered.Though striving to address the entire value chain, the consortium had to operate a selection of priority topics and sub-topics to be tackled throughout the consultation. Hence there is scope for further exploring certain areas in greater detail. Furthermore, workshops are limited to a certain number of participants, and not every invitee was available on the dates set for each workshop. Finally the identification of relevant participants proved to be challenging in fields where networks have not yet been established (this was particularly the case for therapeutic vaccines). As a mitigation strategy, an open public consultation and web-based platform was created to garner further feedback and validate the findings in the roadmap.
Other limitations, due to resource limitations, relate to the fact that IPROVE could not extensively cover in-depth consultations on key areas representing framework conditions that are enablers of innovation, such as in the regulatory and financing domains. These require further reflection and research to understand how to better link the regulatory and science agenda as well as the industrial dimension, as the three are strictly intertwined. Lastly, due to the same challenges described above, IPROVE could not get into the details of a technical implementation plan of the vision set out in this roadmap document. The European Commission and other national stakeholders responsible for incentivising the R&I of vaccines are invited to continue supporting the IPROVE network in coordinating such implementation through their programmes and policies.
2 Plotkin S (2008), Vaccines, Elsevier Health Sciences
3 Rappuoli R, (2014) Presentation at the New Horizon for Vaccine R&D&I in Europe Conference, March 2014
4 Pronker E et al (2013), Risk in Vaccine Research and Development Quantified
5 European Framework Programme 7 funding scheme, dedicated to actions that cover not the research itself, but the coordination and networking of projects, programmes and policies
Source: EC research and innovation, FP7 in brief. https://ec.europa.eu/research/fp7/understanding/fp7inbrief/funding-schemes_en.html
6 O’Neill AMR Review (2016), Vaccines and Alternative Approaches: Reducing our dependence on Antimicrobials, available at www.amr-review.org
7 Vaccines Europe members’ figures for the year 2014. Accessible at www.vaccineseurope.eu
8 http://www.dcvmn.org/sites/default/files/files/DCVMN_jan_2013.pdf Overview of DCVMN capabilities; Dr. Suresh Jadhav Dr. Suresh Jadh “Opportunities for emerging vaccine markets” http://ec.europa.eu/research/health/pdf/event17/s3-10-suresh-jadhav_en.pdf
9 Blank PR et al. (2013), Population access to new vaccines in European countries. Vaccine; 31(27): 2862-7
10 BIOTECanada (2010), Research and Development: Fostering vaccine innovation in Canada
11 IFPMA (2013), Vaccine Research & Development, http://farm9.staticflickr
12 IFPMA (2014), Maintaining the vaccines innovation edge; http://www.ifpma.org/resources/infographics.html
13 WHO (2013), Priority Medicines for Europe and the World (Update), accessible at: http://www.who.int/medicines/areas/priority_medicines/MasterDocJune28_FINAL_Web.pdf
14 The list of IPROVE Advisory Board members is provided at the end of this report
15 More information on the IPROVE Governance structure can be found at www.iprove-eu.eu
16 A full list of all of the stakeholders consulted is provided at the end of this report